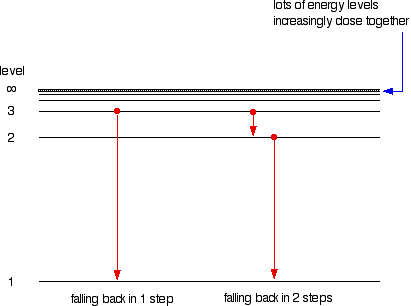
How can we explain the distinct lines that appear in an atomic spectrum quizlet?Įvery element emits a unique line spectrum. Each of these spectral lines corresponds to a different electron transition from a higher energy state to a lower energy state. What makes the spectrum of each chemical element and molecule unique?Īn atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. The lines (photons) are emitted as electrons fall from higher energy orbitals to lower energies. The emission lines correspond to the differences between various pairs of the many energy levels. This occurs because the atom contains specific levels, and an atom can only absorb or emit radiation that corresponds to the energy between these levels.ĭoes each element have a unique emission spectrum?Įach elements emission spectrum is distinct because each element has a different set of electron energy levels. Why does each element have its own unique atomic line spectrum quizlet?Įach element has its own atomic line spectrum, consisting of fine lines of individual wavelengths that are characteristic for the element. It’s a set of frequencies of the electromagnetic spectrum emitted by excited elements of an atom. Because they are unique, they can act as an element s fingerprint. What is a unique atomic emission spectrum?Ītomic emission spectra are unique spectra of light emitted by an element when electricity is run through it or when it is viewed through a prism. Analyzing the colors of light given off by stars reveals which elements are present in those stars. One use of this technique is to identify the elements present in distant stars. The different colors of light produced by emission spectra of different elements allows them to be identified. Why is the atomic emission spectrum important in the real world? The emission spectrum can be used to determine the composition of a material, since it is different for each element of the periodic table. The production of line spectra by the atoms of an element indicate that an atom can radiate only a certain amount of energy. Why is an elements emission spectrum useful? Since each atom has many excited states (high energy levels) available to it, several colors of light can be emitted by each element. Why do all elements have a unique emission spectrum?Īs each element has different energy states available to it, each element releases photons of different color when its atoms return to their lower energy states. 5 How are colors of light used to identify elements?.4 What is a line spectrum in chemistry quizlet?.3 What makes the spectrum of each chemical element and molecule unique?.2 What is a unique atomic emission spectrum?.1 Why do all elements have a unique emission spectrum?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
